Which Best Describes the Structure of an Atom
Rutherfords Atomic Structure. Group of answer choices.
Which best describes the current model of an atom.

. What best describes the nucleus of an atom-. Add an answer or comment. Electrons Compared to the whole atom the nucleus of the atom is.
A nucleus that contains protons neutrons and electrons which are all charged but balance one another B. Neutrons The energy levels around the nucleus of an atom contain. Negative protons and positive neutrons in the nucleus are surrounded by neutral electrons.
A 250-cm-long cylindrical glass tube sealed at one end is filled with ethanol. Which of the following best describes the structure of an atom. An electron has a negative charge and is located outside the nucleus.
Which of these phrases best describes an atom. The shells of an atom are numbered 12 3 and so on starting from the one closest to the nucleus. There are always the same number of protons and neutrons.
Around the year 1911 British physicist Ernest Rutherford carried out the famous Gold foil experiment to understand the structure of an atom. In simple terms an atom is a cloud of tiny electrons buzzing round a central much larger nucleus in a series of orbits called shells. Each shell can occupy a certain number of.
A positively charged nucleus circled by widely spaced negatively charged particles. Neutral protons and negative neutrons in the nucleus are surrounded by positive electrons. His first conclusion was that most of the space inside the atom is empty.
An electron has a positive charge and is located outside the nucleus. Protons have a positive charge neutrons have no charge and electrons have a negative charge. Vastly empty space with a heavy positively charged center.
Thus we can conclude that out of the given options a core of protons and neutrons surrounded by electrons best describes an atom. The density of ethanol is 0789 gmL. The structure of an atom includes which of the following.
Which statement best describes the electronic charges and location of the subatomic particles in an atom. The planets describe the size of the nucleus of the atom compared to the whole atom. Calculate the inner diameter of the tube in centimeters.
A sphere made up of an equal number of protons neutrons and. A positive nucleus surrounded by a cloud of negative charges Most of the volume of the atom is composed of. The Electronic Structure of an Atom.
Log in for more information. 1Which of the following best describes an atom. There are always the same number of protons and electrons.
There are no comments. Protons and neutrons are in the nucleus with electrons orbiting the nucleus. - accurately describes the structure of an atom.
Protons have positive charges located in the nucleus. Electrons have neutral charges located in the electron cloud. A nucleus with negatively charged protons and uncharged neutrons with orbiting positively charged electrons.
Examine the structure of this atom. Charge and mass evenly scattered throughout a spherical shape. Neutrons have negative charges located in the nucleus.
Which of the following best describes the structure of an atom. Lightweight positively charged pieces surrounding a heavy negatively charged center. Electrons are arranged around the nucleus in the shells of an atom.
An atom consists of three sub atomic particles which are protons neutrons and electrons. Which statement describes the charge and location of an electron in an atom. Answer choices a solid sphere with electrons and protons embedded a solid sphere unique for everything that exists a central nucleus containing protons and neutrons with electrons orbiting in levels of high probability a central nucleus containing protons with electrons orbiting in specific paths.
Added 852019 113314 AM. This answer has been confirmed as correct and helpful. The mass of ethanol needed to fill the tube is found to be 4523 g.
There are always the same number of neutrons and electrons. Electrons in set orbits. Positive protons and negative electrons in the nucleus are surrounded by neutral neutrons.
Which correctly describes the structure of an atom. A nucleus that is mostly empty space with charged particles circling around it. Smaller and contains little of the atoms mass.
An electron has a positive charge and is located in the nucleus. A nucleus made up of protons surrounded by an electron cloud. Which of these describes the structure of an atom.
A protons and electrons grouped together in a random pattern B protons and electrons grouped together in an alternating pattern C a core of protons and neutrons surrounded by electrons D a core of electrons and neutrons surrounded by protons. The current model of an atom is best described by the Solar System. A neutral nucleus circled by particles with positive and negative charges.
In the experiment he used a source of alpha particles an atom and a detector. A nucleus made up of protons and neutrons surrounded by. The number of protons neutrons and electrons is always the same E.

Labeled Parts Of An Atom Diagram Atom Diagram Atom Worksheets
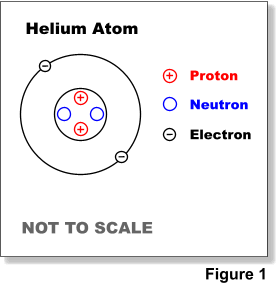
Comments
Post a Comment